Carbon monoxide

Carbon monoxide is a colourless, odourless gas formed when substances containing carbon (such as petrol, gas, coal and wood) are burned with an insufficient supply of air.
Motor vehicles are the main source of carbon monoxide pollution in urban areas. The emissions contain carbon monoxide from the incomplete burning of fuel in the engine.
Health effects
Carbon monoxide has serious health impacts on humans and animals.
When inhaled, the carbon monoxide bonds to the haemoglobin in the blood in place of oxygen to become carboxyhaemoglobin. This reduces the oxygen-carrying capacity of the red blood cells and decreases the supply of oxygen to tissues and organs, especially the heart and brain.
For people with cardiovascular disease, this can be a serious problem.
However, most people in the community need to be exposed to a level of 200 parts per million (ppm) for several hours before they start to feel any effects.
The effects are reversible, so symptoms decrease gradually when exposure to carbon monoxide stops.
Air quality guidelines
The Environmental Protection (Air) Policy 2019 (EPP Air) objective for carbon monoxide is 9ppm for an 8-hour exposure period.
The National Environment Protection (Ambient Air Quality) Measure standards for carbon monoxide is 9ppm for an 8-hour exposure period.
These guidelines have been set to protect the most sensitive members of the community.
Carbon monoxide concentrations measured outdoors in South East Queensland are well below the 8-hour guideline
While ambient carbon monoxide levels in urban residential areas are unlikely to cause adverse health effects, indoor sources, such as poorly ventilated gas appliances and wood heaters, can pose a risk to human health.
Measuring carbon monoxide
We measure carbon monoxide by a technique known as 'gas filter correlation'.
The animated illustration shows how the analyser works.
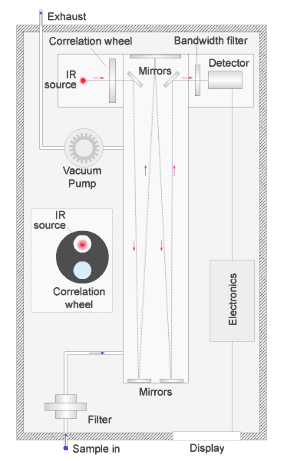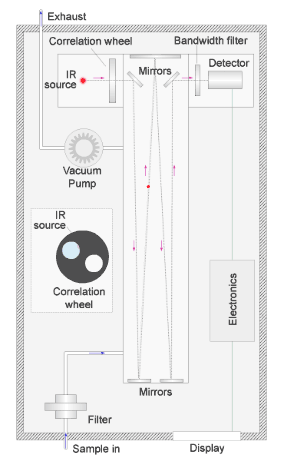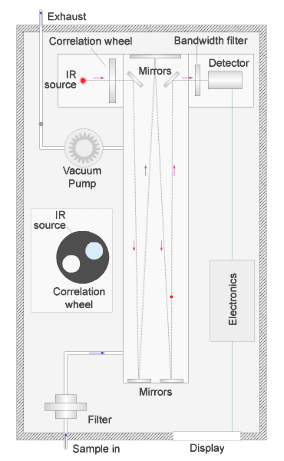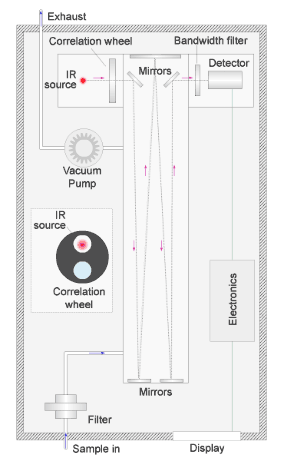
A vacuum pump draws a sample of air through the instrument, shown by the path of the blue dot in the illustration. The sample passes through a filter to remove particles and into a chamber.
In the chamber, a series of mirrors reflect a beam of infrared light back and forward to increase the effective path length, and thus the sensitivity of the measurement.
The red dot in the illustration shows the path of the light. Any carbon monoxide present in the sample absorbs infrared radiation at a wavelength of around 4.7µm (micron) as the beam passes through the chamber.
The beam passes alternately through a reference cell containing non-absorbing nitrogen gas, and a sample cell containing a known air and carbon monoxide mix mounted in a rotating wheel.
The 2 cells pass in front of the infrared source alternately as shown in the simulation of the rotating wheel in the diagram.
The detector measures the difference in the intensity of the infrared light due to absorption by carbon monoxide in the sample cell and the nitrogen-filled reference cell.
The difference allows us to calculate the concentration of carbon monoxide in the air in the chamber.
More information
Go to the live air data service to check the current levels of carbon monoxide in the monitoring network.