Meteorological factors
Air movements influence the fate of air pollutants. So any study of air pollution should include a study of the local weather patterns (meteorology).
If the air is calm and pollutants cannot disperse, then the concentration of these pollutants will build up. On the other hand, when strong, turbulent winds blow, pollutants disperse quickly, resulting in lower pollutant concentrations.
Meteorological data helps:
- identify the source of pollutants
- predict air pollution events such as inversions and high-pollutant concentration days
- simulate and predict air quality using computer models.
When studying air quality, it is important to measure the following factors as they can help us understand the chemical reactions that occur in the atmosphere:
Wind speed and direction
When high pollutant concentrations occur at a monitoring station, wind data records can determine the general direction and area of the emissions. Identifying the sources means planning to reduce the impacts on air quality can take place.
An instrument called an anemometer measures wind speed. At our monitoring stations, the type of anemometer we use is a sonic anemometer.
A sonic anemometer operates on the principle that the speed of wind affects the time it takes for sound to travel from one point to another. Sound travelling with the wind will take less time than sound travelling into the wind. By measuring sound wave speeds in 2 different directions at the same time, sonic anemometers can measure both wind speed and direction.
Temperature
Measuring temperature supports air quality assessment, air quality modelling and forecasting activities.
Temperature and sunlight (solar radiation) play an important role in the chemical reactions that occur in the atmosphere to form photochemical smog from other pollutants.
Favourable conditions can lead to increased concentrations of smog.
The most common way of measuring temperature is to use a material with a resistance that changes with temperature, such as platinum wire. A sensor measures this change and converts it into a temperature reading.
Humidity
Like temperature and solar radiation, water vapour plays an important role in many thermal and photochemical reactions in the atmosphere. As water molecules are small and highly polar, they can bind strongly to many substances. If attached to particles suspended in the air they can significantly increase the amount of light scattered by the particles (measuring visibility). If the water molecules attach to corrosive gases, such as sulfur dioxide, the gas will dissolve in the water and form an acid solution that can damage health and property.
Water vapour content of air is reported as a percentage of the saturation vapour pressure of water at a given temperature. This is the relative humidity. The amount of water vapour in the atmosphere is highly variable—it depends on geographic location, how close water bodies are, wind direction and air temperature. Relative humidity is generally higher during summer when temperature and rainfall are also at their highest.
Measuring humidity uses the absorption properties of a polymer film. The film either absorbs or loses water vapour as the relative humidity of the ambient air changes. A sensor measures these changes and converts them into a humidity reading.
Rainfall
Rain has a 'scavenging' effect when it washes particulate matter out of the atmosphere and dissolves gaseous pollutants. Removing particles improves visibility. Where there is frequent high rainfall, air quality is generally better.
If the rain dissolves gaseous pollutants, such as sulfur dioxide, it can form acid rain resulting in potential damage to materials or vegetation.
A common method to measure rainfall is to use a tipping bucket rain gauge—see illustration.
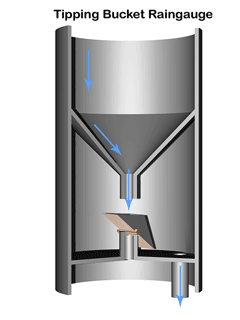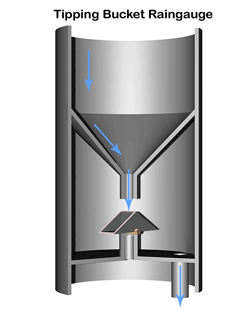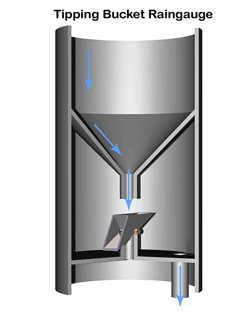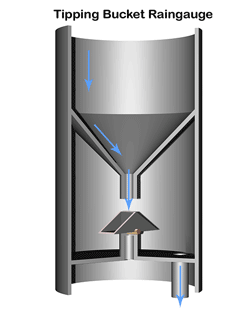
The gauge registers rainfall by counting small amounts of rain collected. When rain falls into the funnel, it runs into a container (the tipping bucket) divided into 2 equal compartments by a partition.
The design of the tipping bucket makes one compartment tilt downward and rest against a stop when it is empty, positioning the other compartment under the funnel ready to receive rain water.
When a set amount of rain has drained from the funnel into the upper compartment the bucket tilts the opposite way so that the compartment containing the rain comes to rest against the stop on the opposite side. The collected water then empties out and the other compartment starts to fill.
The instrument calculates the quantity and intensity of rainfall using with the area of the funnel and the number and rate of bucket movements.
Solar radiation
It is important to monitor solar radiation for use in modelling photochemical smog events, as the intensity of sunlight has an important influence on the rate of the chemical reactions that produce the smog. The cloudiness of the sky, time of day and geographic location all affect sunlight intensity.
An instrument called a pyranometer measures solar radiation from the output of a type of silicon cell sensor.